Information for patients prescribed Nucala (mepolizumab)
This page is specifically for patients who have been prescribed Nucala. If you are not a patient who has already been prescribed Nucala, please return to the public website.

So...you've been prescribed Nucala for severe refractory eosinophilic asthma?
This page is here to help you to understand more about your condition and your Nucala medication. If you have additional questions, ask your doctor, nurse or pharmacist and read the patient information leaflet (PIL) provided with your medicine.
Research has shown that not all asthma is the same. In fact, advances in science have led doctors to realise that different types of asthma require different types of treatment.
Around 4% of adults with asthma have severe asthma.1
Some diagnosed with severe asthma have a particular type: Severe Eosinophilic Asthma.
What makes asthma severe?

One of the ways that asthma can differ is in its severity. In general, patients with severe asthma find it difficult to control their daily asthma symptoms.

Severe asthma patients may continue to have asthma attacks, even when they are taking high doses of prescribed asthma medications or are adherent to treatment.
What are eosinophils?
Eosinophils are a type of white blood cell found throughout the body that play an important role in the immune system.
Some people with severe asthma have too many eosinophils in the blood and lungs, causing the airways to become inflamed, increasing the risk of an asthma attack. This condition is called eosinophilic asthma.
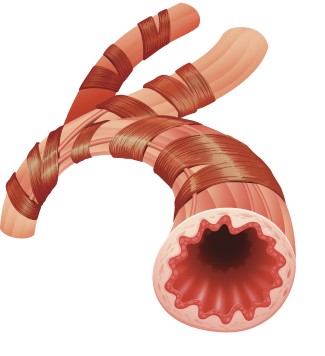
Healthy airway
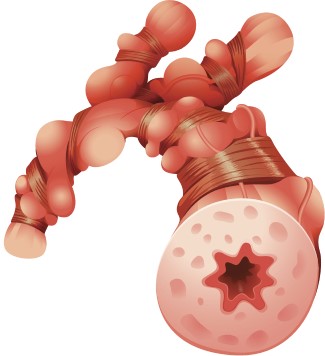
Inflamed airway

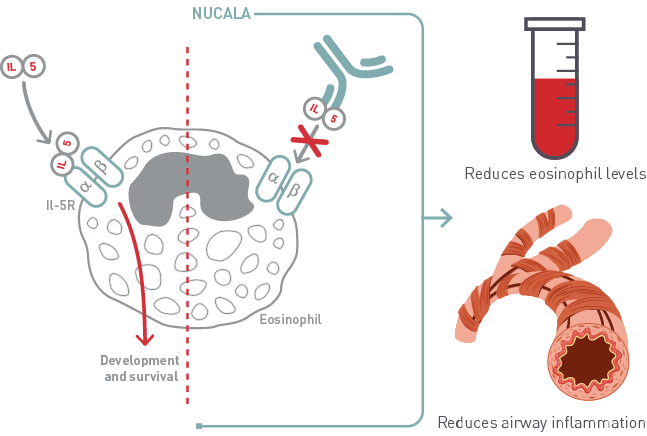
- Nucala is designed to block the activity of a molecule called interleukin-5, or IL-5 for short. IL-5 is a molecule in our body that sends out signals to help eosinophils grow and move around the body.
- Nucala blocks IL-5 binding to eosinophils and reduces the number of eosinophils in your blood, leading to less inflammation in your lung.

Your current asthma symptoms, including the number of asthma attacks

The types of medications you take

Your routine blood tests
What's different about my new treatment?
Nucala is an add-on treatment to your other asthma medications.
It is not an inhaler or an oral tablet. It is given as an injection under the skin (subcutaneous) every 4 weeks.
Asthma Action Plan
- All asthma patients should have an asthma action plan. Download an asthma action plan here and fill it in with your doctor, nurse or pharmacist: https://www.asthmaandlung.org.uk/conditions/asthma/your-asthma-action-plan (this site is not owned or controlled by GSK).
- Keep a written record of your progress to share with your doctor, nurse or pharmacist.
Should I continue my existing medication?
● If you are transitioning to Nucala from another biologic medication for severe asthma, please follow the guidance provided by your doctor, nurse, pharmacist. They will advise you on the appropriate steps.
●Keep taking your other asthma medicines unless your doctor tells you otherwise. Nucala is not a rescue treatment for sudden breathing problems.
● It is recommended that all patients with asthma should have an asthma action plan.
● If you have any questions, please ask your doctor,
nurse or pharmacist at your next clinic appointment.

When can I stop this treatment?
- Do not stop injections of Nucala unless your doctor, nurse or pharmacist advises you to. Interrupting or stopping the treatment with Nucala may lead to your asthma becoming less controlled.
- Nucala is intended for long-term treatment of severe asthma.
- Nucala is an add-on treatment which means that you should continue to take your other asthma medication as prescribed unless advised otherwise by your doctor, nurse or pharmacist.
- Nucala is not a rescue treatment for sudden breathing problems or acute asthma attacks.
- If your asthma symptoms get worse while receiving this medication please seek medical attention.

It’s important to note:
Always consult your doctor, nurse or pharmacist and let them know if...
●Your asthma remains uncontrolled
● Your symptoms worsen
● You are allergic to mepolizumab or any of the other ingredients
● You are breast-feeding
● You are pregnant, think you may be pregnant or are planning to have a baby
● You have any concerns about your treatment
● You are taking any other medication
What are the side effects?
Like all medicines, this medicine can cause side effects, although not everybody gets them. The side effects caused by Nucala are usually mild to moderate but can occasionally be serious. If you’re worried about any symptoms, talk to your doctor, nurse or pharmacist. For the full list, please read the Patient Information Leaflet.
Allergic Reactions
Some people may have allergic or allergic-like reactions.
These reactions may be common (affect between 1 in 100 people to 1 in 10 people). Anaphylaxis (severe allergic reaction) is rare but needs urgent medical attention.
They usually occur within minutes to hours after the injection, but sometimes symptoms can start up to several days later.
If you experience an allergic reaction, please seek medical attention immediately.
Allergic reaction symptoms include:
● Chest tightness, coughing and difficulty breathing
● Fainting, feeling dizzy or lightheaded (due to a drop in blood pressure)
● Swelling of eyelids, face, lips, tongue or mouth
● Hives
● Rash
If you have had a similar reaction to any injection or medicine, tell your doctor before you are given Nucala.
Other side effects include:
Very Common side effects
These may affect more than 1 in every 10 people taking Nucala, include:
- Headache
Common side effects
These may affect 1 in 100 to 1 in 10 people taking Nucala, include:
- Chest infection - symptoms of which may include cough and fever (high temperature)
- Fever (high temperature)
- Eczema (itchy red patches on the skin)
- Urinary tract infection (symptoms may include painful and frequent urination, fever, pain in lower back, cloudy or bloody urine)
- Upper abdominal pain (stomach pain or discomfort in the upper area of the stomach / abdomen)
- Injection-site reaction (pain, redness, swelling, itching, and burning sensation of the skin near where the injection was given)
- Back pain
- Arthralgia (joint pain)
- Pharyngitis (sore throat)
- Nasal congestion (stuffy nose)
- Systemic non-allergic administration-related reactions such as rash, flushing, myalgia and fatigue
- Herpes zoster*
*Herpes zoster was reported uncommonly in severe asthma studies.
Rare side effects
These may affect 1 in 10,000 to 1 in 1,000 people taking Nucala, include:
Anaphylaxis (severe allergic reaction)
Please tell your doctor, nurse or pharmacist immediately if you experience any of these side effects.
Reporting of Side Effects
If you get any side effects, talk to your doctor or nurse. This includes any possible side effects not listed above.
You can also report side effects directly via the Yellow Card Scheme at: https://yellowcard.mhra.gov.uk/ or search for MHRA Yellowcard in the Google Play or Apple App store. Adverse events should also be reported to GlaxoSmithKline on 0800 221 441.
By reporting side effects you can help provide more information on the safety of this medicine.
Things to remember 2
- Do not inject Nucala before you or your caregiver have received training on how to use Nucala.
- Do not shake the pen.
- Do not use the pen if dropped onto a hard surface.
- Do not use the pen if it appears damaged.
- Do not remove the needle cap until just before your injection.
- Do not freeze the pen.
- Do not use if the expiry date (which is stated after 'EXP') has passed. The expiry date on your pen refers to the last day of that month.
- Do not inject if the solution looks cloudy or discoloured, or has particles.
- Do not share your Nucala pre-filled pen with another person.
Keep Nucala and all medications out of the sight and reach of children.
How to store your pen:
- Store in the refrigerator between 2°C to 8°C.
- Keep in the original carton until time of use to protect from light.
- If necessary, the pen can be removed from the refrigerator and kept in the unopened pack for up to 7 days at room temperature (up to 30°C), when protected from light. The pen should be discarded if left out of the refrigerator for more than 7 days.
- The pre-filled pen should be used only once and then discarded.
- Nucala pre-filled pen is for use under the skin only (subcutaneous).
References
- Asthma+Lung UK. (2026) What is severe asthma? Available at: https://www.asthma.org.uk/advice/severe-asthma/what-is-severe-asthma/ (Last accessed: February 2026) This site is not owned or controlled by GSK.
- Nucala Patient information leaflet
Nucala is a registered trademark of the GSK group of companies


